| Table of Contents |  |
|
Case Report
| ||||||
| Bilious pleural effusion from metastatic serous adenocarcinoma of the ovary: A case report | ||||||
| Hanna Ellingsen1, Ashley N. Elsensohn2, Vijal Patel2, Samantha Flynn2 | ||||||
|
1MS4, Sidney Kimmel Medical College, Thomas Jefferson University, Philadelphia, PA.
2Department of Internal Medicine, Lankenau Medical Center, Wynnewood, PA (US). | ||||||
| ||||||
|
[HTML Abstract]
[PDF Full Text]
[Print This Article]
[Similar article in Pumed] [Similar article in Google Scholar] |
| How to cite this article: |
| Ellingsen H, Elsensohn AN, Patel V, Flynn S. Bilious pleural effusion from metastatic serous adenocarcinoma of the ovary: A case report. J Case Rep Images Med 2016;1:5–8. |
|
Abstract
|
|
Introduction:
The pleural cavity is the most common extra-abdominal site for ovarian cancer metastasis. It can, however, be difficult to diagnose ovarian malignancy from the presence of a pleural effusion alone.
Case Report: A 50-year-old premenopausal woman who presented with a large, right-sided, bilious pleural effusion subsequently diagnosed with advanced ovarian cancer. Conclusion: Metastatic ovarian carcinoma should be considered in appropriate populations when another, more obvious source of the pleural effusion is not evident. In bilious effusions, an overt tract may not be visualized despite appropriate diagnostic imaging and gastrointestinal studies. In these cases, video-assisted thoracoscopic surgery (VATS) can provide a definitive diagnosis and potential therapeutic interventions by selecting patients who could benefit from surgical and chemotherapeutic interventions. | |
|
Keywords:
Advanced ovarian cancer, Bilious pleural effusion, Malignant pleural effusion, Metastatic effusion
| |
|
Introduction
| ||||||
|
The pleural cavity is the most common extra abdominal site for metastatic ovarian cancer [1]. Pleural effusions may be the initial presentation of malignancies, with common origins including carcinomas of the lung, breast or ovary, or lymphomas. Malignant pleural effusions are associated with poor prognosis, the most common histologic subtype being metastatic adenocarcinoma, [2] [3]. Our case is that of a 50-year-old premenopausal woman who presented with a large, bilious pleural effusion. After extensive workup, she was found to have malignant serous adenocarcinoma of the ovary that had metastasized to the pleural space. We review the diagnostic challenges, considerations and subsequent tools for the diagnosis and management of malignant pleural effusion from advanced ovarian cancer. | ||||||
|
Case Report
| ||||||
|
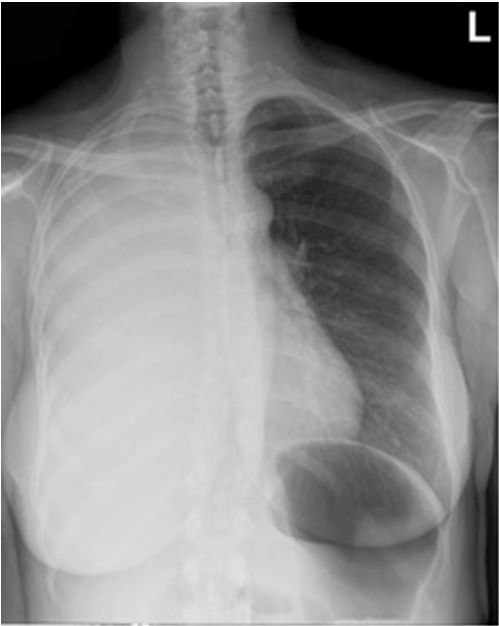
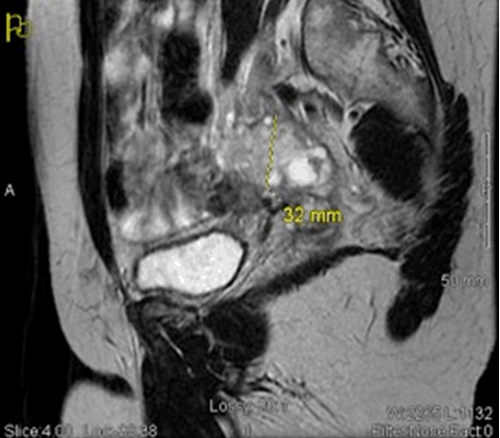
A 50-year-old pre-menopausal female with a history of laparoscopic cholecystectomy for chronic cholecystitis presented with an eight-month history of cough and shortness of breath. She had undergone several outpatient treatments for suspected asthma and bronchitis with inhaled bronchodilators, corticosteroids and azithromycin that failed to improve her symptoms. Just prior to presentation, she had an outpatient chest X-ray performed which demonstrated a large right-sided pleural effusion. She had a history of tuberculosis exposure, but subsequent PPD tests were negative. Patient denied fever, chills, recent weight loss, and lymphadenopathy. On examination, the patient was afebrile, normotensive, without tachypnea or tachycardia. Oxygen saturation was 100% on room air. There were markedly decreased breath sounds in the right lung with dullness to percussion. There were no rhonchi, rales, or wheezing. The remainder of the physical examination was unremarkable. Chest X-ray demonstrated complete opacification of the right hemithorax, indicative of a pleural effusion (Figure 1). Routine labs including a complete blood count and complete metabolic panel were within normal limits. Serum CA-125 was 699 U/ml (normal 0-35 U/ml). Serum rheumatoid factor was < 20.0 IU/ml (significant if more than 150 IU/ml). A thoracentesis yielded 650 cc of opaque, green-tinged pleural fluid. Pleural fluid analysis demonstrated an exudative process. The pH was 7.53, glucose 2 mg/dl, protein 9.0 g/dl and protein ratio of 0.8, cholesterol 152 mg/dl, triglycerides 76 mg/dl, bilirubin 3.1 mg/dl, LDH 1433 U/L, and amylase 439 U/L. Serum alkaline phosphatase was 62 U/L (range 35–126 U/L), LDH 200 U/L (range 98–192 U/L). Pleural fluid cell counts were elevated with red blood cells 17000 (range <10,000 cells/mm3) and WBC 1128 cells/mm3 (normal range < 200 cells/mm3), with a 89% neutrophil predominance. Cultures and stains were negative for bacteria, fungus, and tuberculosis. Differential diagnosis of green pleural exudate in our patient included bilious effusion secondary to previous cholecystectomy, tuberculosis, metastatic gastrointestinal malignancy, metastatic ovarian malignancy, and rheumatoid pleurisy [1]. Given the bilious effusion and elevated amylase, there was concern for biliary leak. HIDA and ERCP were unrevealing. An MRI scan of abdomen did not show any abnormalities of the liver or biliary ducts. MRI scan of pelvis showed mild ascites and enlargement of the ovaries with soft tissue enhancement (Figure 2). This, in conjunction with elevated CA-125, raised concern for ovarian malignancy. Gynecology-oncology, pulmonology, cardiothoracic surgery, general surgery, and gastroenterology were consulted. The patient underwent video-assisted thoracoscopy (VATS) of the right lung with evacuation and subsequent biopsy of the right pleura. Histological examination showed metastatic serous adenocarcinoma consistent with a primary ovarian tumor. The tumor cells stained positive for CK7, CA-125 and D2-40. Following diagnosis, the patient was discharged in stable condition with plans to follow up with gynecology-oncology and pulmonology as an outpatient for further staging and treatment. She elected to follow-up elsewhere, and the details of her ongoing care are unknown. | ||||||
| ||||||
| ||||||
|
Discussion
| ||||||
|
Diagnosing metastatic cancer on the basis of pleural effusion alone can be challenging. As in our case, pleural effusion can be one of the only presenting signs of a malignancy. In a retrospective study of 123 women with malignant pleurisy, effusion was the presenting manifestation of cancer in 29% (36) [3]. Another retrospective study reviewed 742 malignant pleural effusions [4]. Top etiologies were lung (273 patients, 37%), breast (127, 17%), hematologic (74, 10%), and ovarian cancer (50, 7%). More than two-thirds of effusions were large, occupying half or more of the hemithorax. Part of the diagnostic challenge of such cases is that the differential diagnosis for pleural effusions is vast. It includes, but is not limited to, cirrhosis, heart failure, tuberculosis and other infectious etiologies, constrictive pericarditis, endometriosis, and malignancy [1]. Of interest, our patient presented with an exudative bilious effusion. This led to procedures and imaging looking for a biliary tract fistula- workup included a HIDA, abdominal CT scan, MRI scan, and ERCP, none of which showed an identifiable tract. With no biliary source, rheumatoid factor was ordered. Green-yellow effusions can be seen with rheumatoid pleurisy and occur in about 2–3% of patients with rheumatoid arthritis (RA) [5]. This effusion is typically exudative with a high RF titer; rarely, rheumatoid effusions can have features of a sterile empyematous exudate with high lipids and lactate dehydrogenase, and very low glucose and pH levels. Approximately, 75% of patients with ovarian cancer are diagnosed at advanced stages, either Stage III or Stage IV [6]. Stage IV ovarian cancer, diagnosed in our patient, occurs when cancer has spread to the liver parenchyma or outside the abdomen. The most common extra-abdominal site of disease is the pleural surface, with effusions present in greater than one-third of stage IV diagnoses [7]. Malignant pleural effusions in ovarian cancer result from pleural invasion from nearby structures, such as the diaphragm or other pleuroperitoneal communications [8] . These can be difficult to detect with standard lab and imaging modalities, as mentioned above. Certain radiological findings are more predictive of a malignant effusion in those with known ovarian cancer. The predictors of malignant pleuritis are effusions of moderate to large size (81% versus 9% of those studied), supra-diaphragmatic lymph node enlargement of >1 centimeter (75% versus 9%) and pleural nodules of >3 millimeters (50% versus 0%) [9]. Making diagnosis more challenging, effusion cytology did not provide us with a definitive diagnosis. About 30% of malignant pleural effusions from ovarian cancer exhibit false-negative cytological pleural fluid results [1]. In these circumstances, exploratory video-assisted thoracoscopic surgery (VATS) can be used as a diagnostic, staging, and therapeutic modality. A retrospective case review by Diaz et al. reviewed patients with untreated ovarian cancer and moderate to large pleural effusion who underwent VATS. In these patients, VATS revealed macroscopic pleural disease in 29 (69%), the majority (18 patients) having nodules greater than 1 cm [10]. The standard treatment of advanced epithelial ovarian cancer includes surgery followed by adjuvant systemic chemotherapy [11]. Complete resection in stage IV patients taken to primary surgery is about 10%. The goal of surgical cytoreduction is to have no visible residual disease in any location. Based on the retrospective study above, in those with VATS findings, management plans were changed in 18 (43%) patients- 6 underwent intrathoracic cytoreduction first followed by abdominal surgery on the same or a later day; and 12 received neoadjuvant chemotherapy for unresectable pleural tumors followed by interval debulking [10]. The former showing the importance of VATS in influencing the primary management choice in patients with newly diagnosed advanced ovarian cancer that has spread to the pleural space. | ||||||
|
Conclusion
| ||||||
|
It can be difficult to diagnose metastatic ovarian cancer from a presentation of pleural effusion alone. A high degree of suspicion must be maintained and a combination of diagnostic tools may be employed, including labs like CA-125, magnetic resonance imaging scan of abdominal and pelvic, and video-assisted thoracoscopic surgery (VATS). In bilious effusions, an overt tract may not be visualized even with extensive imaging and gastrointestinal studies. VATS can provide more definitive tissue diagnosis with potential for therapeutic interventions by selecting patients who can benefit from intrathoracic cytoreduction or neoadjuvant chemotherapy before primary surgery in the abdomen and pelvis. | ||||||
|
References
| ||||||
| ||||||
|
[HTML Abstract]
[PDF Full Text]
|
|
Author Contributions
Hanna Ellingsen – Substantial contributions to conception and design, Acquisition of data, Drafting the article, Revising it critically for important intellectual content, Final approval of the version to be published Ashley Nicole Elsensohn – Substantial contributions to conception and design, Analysis and interpretation of data, Drafting the article, Revising it critically for important intellectual content, Final approval of the version to be published Vijal Patel – Acquisition of data, Analysis and interpretation of data, Revising it critically for important intellectual content, Final approval of the version to be published Samantha Flynn – Analysis and interpretation of data, Revising it critically for important intellectual content, Final approval of the version to be published |
|
Guarantor of submission
The corresponding author is the guarantor of submission. |
|
Source of support
None |
|
Conflict of interest
Authors declare no conflict of interest. |
|
Copyright
© 2016 Hanna Ellingsen et al. This article is distributed under the terms of Creative Commons Attribution License which permits unrestricted use, distribution and reproduction in any medium provided the original author(s) and original publisher are properly credited. Please see the copyright policy on the journal website for more information. |
|
|