|
Case Series
Three cases of neuropsychiatric systemic lupus erythematosus treated with telitacicept
1 Department of Rheumatology and Immunology, Clinical Medical College, The Affiliated Hospital of Southwest Medical University, Luzhou, China
2 Department of Rheumatology and Immunology, Sichuan Academy of Medical Sciences and Sichuan Provincial People’s Hospital, Chengdu, China
Address correspondence to:
Xixi Chen
Li Long, No. 32, West Section 2, 1st Ring Road, Qingyang District, Chengdu, Sichuan,
China
Message to Corresponding Author
Article ID: 100085Z09WZ2026
Access full text article on other devices

Access PDF of article on other devices

How to cite this article
Zhang W, Zheng Y, Chen X, Long L. Three cases of neuropsychiatric systemic lupus erythematosus treated with telitacicept. J Case Rep Images Med 2026;11(1):5–11.ABSTRACT
Introduction: Neuropsychiatric lupus (NPSLE) is a serious complication of systemic lupus erythematosus (SLE). Telitacicept, a novel B-cell-targeting biologic, has been approved in China for SLE treatment; however, evidence supporting its use specifically in NPSLE remains limited. Evidence for telitacicept in NPSLE is currently limited to individual case reports. As one of the first case series to address this issue, we summarize the treatment courses of these patients to provide stronger evidence for clinical practice.
Case Series: This retrospective case series describes three patients with NPSLE who had an insufficient response to conventional immunosuppressants. Follow-up periods ranged from 7 to 36 months. After adding combination therapy with telitacicept (160 mg weekly), all patients exhibited significant improvement in neuropsychiatric symptoms, a marked reduction in disease activity score, and amelioration in relevant immunological and imaging parameters. No serious adverse events were observed during follow-up.
Conclusion: Telitacicep may offer therapeutic benefit in managing NPSLE, particularly in cases presenting with predominant psychiatric manifestations.
Keywords: Biologics, Neuropsychiatric systemic lupus erythematosus, Telitacicept, Treatment
Introduction
Systemic lupus erythematosus (SLE) is a complex autoimmune disease. When it involves the central or peripheral nervous system, it is classified as neuropsychiatric SLE (NPSLE), which can present as the initial or sole clinical manifestation of SLE [1]. Reported prevalence rates for NPSLE vary widely, ranging from 37% to 95%, which usually correlates with SLE activity [2].
Telitacicept is a new fusion protein drug that binds to B-lymphocyte stimulating factor (BLyS) and apical protein-related inducible ligand (APRIL). It stops the development and survival of plasma cells and mature B cells to achieve treatment effects. This drug was approved by China’s National Medical Products Administration (NMPA) in 2021 for treating patients with active SLE [3]. But in the Phase IIb and Phase III clinical trials of telitacicept, these trials mainly looked at overall results for SLE. They did not do a special data study for NPSLE [4]. This paper reports the clinical data of three NPSLE patients treated at Sichuan Provincial People’s Hospital. It aims to explore the therapeutic efficacy and application prospects of telitacicept in this specific patient group.
CASE SERIES
Case 1
Baseline Characteristics
A 29-year-old female presented with recurrent edema in the lower limbs and face. Eleven years prior to admission, she was evaluated at another hospital for similar symptoms. Initial urinalysis showed 3+ proteinuria, leading to a diagnosis of systemic lupus erythematosus (SLE) with lupus nephritis. Her symptoms resolved after treatment with glucocorticoids and hydroxychloroquine. She subsequently discontinued all medications and remained clinically stable for six years. One year before the current admission, her symptoms recurred. She was then treated with prednisone, hydroxychloroquine, and mycophenolate mofetil, although she later self-tapered these medications. Her condition remained generally stable during this period.
Neuropsychiatric Manifestations
Six days prior to admission, she developed a fever. On the sixth day of hospitalization, her condition deteriorated rapidly. She presented with headache, nausea, vomiting, and persistent high fever. Her symptoms quickly progressed to delirium, slowed responses, drowsiness, and frequent seizures.
Diagnostic Workup
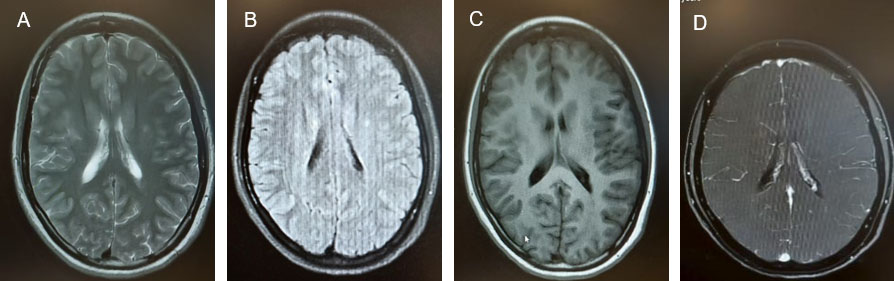
Serological and immunological testing revealed positive antinuclear antibodies (ANA 1:320), anti-Smith antibodies (+), anti-nRNP/Sm antibodies (+), and hypocomplementemia (C3 0.576 g/L, C4 0.174 g/L). Pancytopenia was observed (WBC 2.24 × 109/L, Hb 92 g/L, PLT 88 × 109/L), and the erythrocyte sedimentation rate (ESR) was elevated at 118 mm/h. The anticardiolipin antibody level was 9.28 RU/mL. Multiple autoantibodies were positive, including anti-PCNA, anti-PM-Scl, anti-Ku, and anti-neuronal antibodies. Multiple cerebrospinal fluid (CSF) analyses were performed (Table 1); all tests for infectious etiologies were negative. Contrast-enhanced brain magnetic resonance imaging (MRI) revealed multiple intracranial abnormal signals (Figure 1).
Based on the cumulative CSF findings, lack of response to antimicrobial therapy, and imaging results, evidence for intracranial infection was considered insufficient. According to the 2019 EULAR/ACR classification criteria for SLE and the 1999 ACR criteria for neuropsychiatric lupus, the patient was diagnosed with NPSLE manifesting as acute confusional state and seizures, with concurrent hematologic involvement. Her SLE Disease Activity Index (SLEDAI-2K) score was 29.
Treatment Timeline
Upon the onset of neuropsychiatric symptoms, the patient received methylprednisolone pulse therapy (1 g/day for 5 days, tapered to 500 mg/day for 5 days) in combination with intravenous immunoglobulin (IVIG, 17.5 g/day for 5 days). Hydroxychloroquine (0.2 g twice daily) was continued. Levetiracetam (0.5 g twice daily) and perampanel (2 mg once nightly) were initiated for seizure control. Following high-dose corticosteroid and IVIG therapy, her fever peaks subsided and seizure frequency decreased. However, she remained somnolent and experienced occasional myoclonic jerks. Repeat laboratory tests showed a persistently elevated ESR (81 mm/h), mild anemia (Hb 92 g/dL). Given the high autoantibody titers and inflammatory markers, treatment with telitacicept (160 mg subcutaneously once weekly) was initiated.
Outcomes with Follow-up
At seven months after treatment initiation, the patient showed significant improvement in neuropsychiatric symptoms. She resumed normal daily functioning, with fluent speech and independent ambulation, although occasional limb twitching persisted. Laboratory findings revealed normalization of complete blood count and complement levels, with mildly reduced IgG and IgM levels. Electroencephalography (EEG) findings remained moderately abnormal. The patient achieved partial remission of NPSLE symptoms. Her maintenance regimen included prednisone (10 mg once daily), hydroxychloroquine (200 mg twice daily), telitacicept (160 mg weekly), sertraline (50 mg once daily), donepezil (10 mg once daily), and levetiracetam (0.5 g twice daily).
Case 2
Baseline Characteristics
A 52-year-old woman presented with neuropsychiatric symptoms. She had initially been managed at our hospital 11 months prior to the current admission, presenting at that time with purpura on both lower limbs. Physical examination then revealed non-blanching purpura scattered over the abdomen and legs. Laboratory tests demonstrated leukopenia (WBC 3.68 × 109/L), mild anemia (Hb 113 g/L), positive antinuclear antibodies (ANA 1:320), and positivity for anti-SSA, anti-Ro-52, anti-nucleosome, and anti-dsDNA antibodies. Elevated inflammatory markers (CRP 29.82 mg/L, ESR 27 mm/h) and decreased complement levels (C3 0.333 g/L, C4 < 0.074 g/L) were also noted. Based on these findings, she was diagnosed with SLE and treated with prednisone, tacrolimus, and hydroxychloroquine.
Neuropsychiatric Manifestations
Fifteen days prior to admission, she developed psychiatric symptoms, including involuntary crying and dysarthria.
Diagnostic Workup
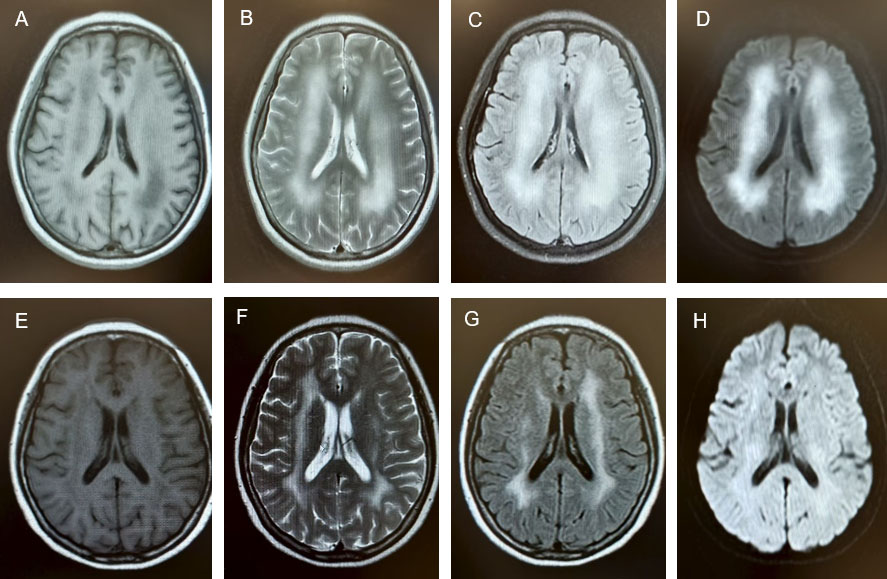
Serological and immunological testing demonstrated persistent autoantibody positivity (ANA 1:320, anti-dsDNA++, anti-AHA+, anti-ANA+, anti-SSA+, and anti-RO52+). The anticardiolipin antibody level was 36.48 RU/mL. Hypocomplementemia (C3 0.367 g/L, C4 < 0.063 g/L) and an elevated ESR (37 mm/h) were also observed. Lumbar puncture revealed elevated cerebrospinal fluid pressure (160 mmH2O) and increased protein concentration (0.39 g/L). Cerebrospinal fluid analysis performed at another hospital had previously shown glucose 8.3 mmol/L, chloride 116 mmol/L, protein 0.31 g/L, and IgG 60.1 mg/L; oligoclonal bands and antibodies against central nervous system demyelination were negative. Brain MRI performed at another hospital disclosed multifocal hyperintensities involving both frontoparietal lobes, the centrum semiovale, and the cerebellomesencephalic regions. Contrast-enhanced brain MRI at our institution demonstrated symmetrical patchy lesions with slightly prolonged T1 and prolonged T2 signals in the bilateral cerebellar middle peduncles, the centrum semiovale, and the periventricular regions of the lateral ventricles. These lesions showed high signal intensity on FLAIR sequences and restricted diffusion on diffusion-weighted imaging (DWI) (Figure 2). Electroencephalography revealed moderate abnormalities. According to the 2019 EULAR/ACR classification criteria for SLE, the patient was diagnosed with SLE with neuropsychiatric involvement, manifesting as mood disorder and possible cognitive dysfunction. Her SLEDAI-2000 score was 20.
Treatment Timeline
The patient was treated with methylprednisolone (40 mg/day for 10 days), cyclophosphamide (200 mg every other day), hydroxychloroquine (200 mg twice daily), and IVIG (12.5 g/day for 5 days). Telitacicept (160 mg weekly) was initiated concurrently with the above immunosuppressive therapy. Following psychiatric consultation, memantine (5 mg once nightly) and donepezil (5 mg once nightly) were prescribed. After two weeks of treatment, the patient occasionally exhibited intense crying and slowed reactions, but maintained clear speech and relevant responses.
Outcomes with Follow-up
At the one-month follow-up, lower limb weakness had improved, with no numbness in the limbs and only occasional involuntary crying. Prednisone was reduced to 40 mg once daily, with a weekly dose reduction of 5 mg. After receiving a cumulative dose of 12 g of cyclophosphamide, treatment was switched to mycophenolate mofetil 500 mg twice daily. At the six-month follow-up, the patient no longer experienced uncontrollable crying and was able to walk independently. Laboratory results showed normalization of hemoglobin (141 g/L) and WBC count (3.31 × 109/L), and complement levels (C3 0.722 g/L, C4 0.146 g/L). Brain MRI revealed patchy areas with slightly prolonged T1 and T2 signals in the periventricular regions of both lateral ventricles and the centrum semiovale, with high signal intensity on FLAIR sequences (Figure 2). No new abnormalities were noted compared with prior imaging. Her SLEDAI-2K score had decreased from 20 at baseline to 2. The patient achieved significant clinical and laboratory improvement, with disease in remission. Her maintenance regimen included prednisone (5 mg weekly), hydroxychloroquine, mycophenolate mofetil, and telitacicept, with regular outpatient follow-up.
Case 3 Baseline Characteristics
A 34-year-old man presented with progressive limb numbness and weakness. He had initially been managed at another hospital six months prior to admission. At that time, physical examination revealed reduced muscle strength in all four limbs and impaired pinprick sensation distally. Laboratory tests demonstrated autoantibody positivity (ANA+, anti-nRNP+, anti-SSA++, anti-Ro52++, anti-sulfatides+). Complement levels were within normal range (C3 0.990 g/L, C4 0.101 g/L). Urinalysis showed 1+ proteinuria, and liver enzymes were elevated (AST 81.3 U/L, ALT 128 U/L, ALP 131 U/L, GGT 98 U/L). Inflammatory markers were modestly elevated (CRP 4.10 mg/L, ESR 45 mm/h). Electromyography and nerve conduction studies indicated peripheral nerve damage. Cerebrospinal fluid analysis showed glucose 3.14 mmol/L, chloride 125 mmol/L, and protein 1.33 g/L; microscopy, culture, and next-generation sequencing (NGS) were unrevealing. Based on these findings, he was diagnosed with Guillain–Barré syndrome. He received IVIG (32 g once daily for 5 days), methylprednisolone (1 g/day for 3 days, tapered to 500 mg/day for 3 days), followed by oral prednisone administered at 30 mg once daily, with the dose tapered by 10 mg weekly until discontinuation. His numbness and weakness in the limbs improved, and he regained the ability to walk independently.
Neuropsychiatric Manifestations
One month prior to admission, the patient experienced recurrent weakness in all four limbs and difficulty walking, accompanied by fever, bilateral knee joint pain, and myalgia. While neuropsychiatric symptoms were not the predominant feature, the recurrent limb weakness with peripheral neuropathy was attributed to NPSLE involvement.
Diagnostic Workup
Serological and immunological testing in outpatients of our hospital revealed positive antinuclear antibodies (ANA 1:100), anti-nRNP/Sm (+), anti-mitochondrial M2 (++), anti-SSA (++), anti-Ro52 (+), and anti-ribosomal P protein antibodies. Both p-ANCA and c-ANCA were detected. The anticardiolipin antibody level was 19.73 RU/mL, and anti-dsDNA was 46.36 IU/mL. Laboratory testing also demonstrated leukopenia (WBC 3.49 × 109/L) and low complement levels (C3 0.605 g/L, C4 < 0.064 g/L). Urinalysis revealed proteinuria (+), with a 24-hour urine protein total of 18.24 g/24 h. Cerebrospinal fluid analysis performed at initial presentation had shown elevated protein (1.33 g/L), with a negative infectious workup. Electromyography and nerve conduction studies from the prior admission indicated peripheral nerve damage. The patient was diagnosed with NPSLE manifesting as peripheral neuropathy, with concurrent hematologic and renal involvement. His SLE Disease Activity Index (SLEDAI-2K) score was 17.
Treatment Timeline
The patient was treated with methylprednisolone pulse therapy (1000 mg/day for three days, tapered to 80 mg/day for four days, and then maintained at 48 mg/day), in combination with hydroxychloroquine (200 mg twice daily), tacrolimus (0.5 mg twice daily), and total glucosides of paeony (600 mg twice daily). Given the patient’s elevated inflammatory markers and high disease activity, telitacicept was initiated at 160 mg once weekly.
Outcomes with Follow-up
At the one-month follow-up, the patient was afebrile, and his joint pain and limb weakness had improved. Laboratory results showed microalbuminuria at 143.60 mg/L, with 24-hour urinary protein excretion reduced to 2.120 g/24 h (improved from 18.24 g/24 h). Complement levels had increased (C3 0.758 g/L, C4 0.094 g/L), and anti-dsDNA antibodies had decreased to 12.93 IU/mL. Prednisone was gradually tapered. After 11 months, 24-hour urinary protein was near normal (0.122 g/24 h). Complement levels had normalized (C3 1.250 g/L, C4 0.253 g/L), and anti-dsDNA antibodies were 2.72 IU/mL. The patient’s SLEDAI-2K score had decreased from 17 at baseline to 2 and prednisone was further reduced to 7.5 mg daily. At the 36-month follow-up, the patient’s condition remained stable, with normal urinary protein, complete blood count, and complement levels (C3 1.500 g/L, C4 0.299 g/L). Prednisone was discontinued, and maintenance therapy with telitacicept (160 mg weekly), hydroxychloroquine (200 mg twice daily), and tacrolimus (0.5 mg twice daily) was continued with regular outpatient follow-up.
Discussion
Neurological involvement was a prominent feature in all three patients: two had central nervous system involvement and one had peripheral nervous system involvement. One patient had their first disease onset, but the other two had confirmed diagnoses before and were in the maintenance phase after induction remission. Besides neurological involvement, other systems were affected, including renal and hematologic involvement. Systemic lupus erythematosus Disease Activity Index scores were over 12 points in all cases. All three patients initially received conventional corticosteroid therapy combined with immunosuppressants, but their symptoms remained uncontrolled with ongoing disease activity. Consequently, the biologic agent telitacicept was added. During outpatient follow-up, all three patients demonstrated significant improvement in symptoms, with reduced neurological, hematological, and renal involvement. Their disease activity scores decreased to below 4.
The central nervous system (CNS) has long been seen as an immune-privileged site. This is mainly because of the blood-brain barrier (BBB). The BBB limits the transport of immune cells and cytokines from the blood to the CNS. Breakdown of the BBB is seen as a key factor in the development of NPSLE [5]. The exact cause of NPSLE is not fully understood yet. However, studies show that B cells, autoantibodies, and cytokines play central roles in its disease process [6].
Abnormal B-cell activation plays a basic role in the disease process of SLE [7]. In NPSLE, cytokines from peripheral or central sources can break down the BBB. This allows autoantibodies and B cells to enter the central nervous system (CNS). This process leads to neurotoxicity and glial cell activation through autoantibody deposition [8].
B-lymphocyte stimulating factor belongs to the tumor necrosis factor (TNF) family and is essential for B-cell maturation. B-lymphocyte stimulating factor binds to receptors on the B cell surface and then promotes B cell activation, growth, and antibody secretion [9]. It can also directly break down the BBB. This worsens the infiltration of autoantibodies and B cells into the CNS [10],[11],[12]. Targeting this pathway has become a promising strategy for NPSLE management. Belimumab is a monoclonal antibody targeting BAFF. It is not approved specifically for NPSLE. But it has shown good effects on neuropsychiatric symptoms in severe NPSLE [13],[14]. Two Phase III trials of belimumab in patients with SLE also showed improvements in CNS-related symptoms. But some case reports said NPSLE patients receiving belimumab had worsening psychiatric symptoms [15].
Apical protein-related inducible ligand is another key molecule in this pathway. It promotes B-cell survival and antibody production in the CNS. This worsens neuronal damage [16]. Apical protein-related inducible ligand levels in CSF are higher in NPSLE patients than in SLE patients without CNS involvement [17]. These findings show that blocking BLyS and APRIL could be treatments for NPSLE.
Telitacicept is a new fusion protein biologic. It blocks the downstream signaling pathways by binding to the common receptor transmembrane activator and calcium modulator and cyclophilin ligand interactor (TACI) of BLyS and APRIL. The medicine stops plasma cells from growing and living and also reduces the production of autoantibodies [18],[19]. Telitacicept has been approved in China for the treatment of highly active SLE that is unresponsive to conventional therapy. Its potential neuroprotective effect provides a new direction for the treatment of NPSLE, and recently, there have been case reports of patients with lupus encephalopathy showing symptom relief after treatment with telitacicept [20].
The most common adverse events associated with telitacicept are respiratory tract infections and injection site infections [4]; no such adverse reactions were recorded in the case series.
This study has some limits. First, the number of patients was small (only three). Also, there was no control group. So it is hard to be sure of the results and to prove cause and effect. Second, all patients used steroids and other immunosuppressant drugs at the same time. So it is hard to tell what effect came from telitacicept alone. Third, the study was done in only one place. So the results may not apply to other hospitals or groups of patients.
Adverse events were checked at each visit through clinical assessments and laboratory tests. These included regular measurements of serum IgG, IgA, and IgM levels and checks for injection site reactions. All three patients were treated with telitacicept 160 mg weekly for 6–36 months. No serious adverse events, infections, or injection site reactions occurred. Immunoglobulin levels decreased gradually. Immunoglobulin G dropped from 11.2–13.5 g/L at baseline to 6.5–8.5 g/L at last follow-up. Immunoglobulin M dropped from 1.5–2.1 g/L to 0.7–0.9 g/L. No liver or kidney toxicity was observed.
Conclusion
These cases suggest that telitacicept may provide a new treatment option for NPSLE. This is especially true for patients with refractory disease or those who cannot tolerate conventional treatments. Its dual block of BLyS/APRIL may more precisely control immune dysregulation. It also shows good safety. More clinical studies are needed to find the best patient group, dosing plan, and long-term efficacy.
REFERENCES
1.
Ameer MA, Chaudhry H, Mushtaq J, Khan OS, Babar M, Hashim T, et al. An overview of systemic lupus erythematosus (SLE) pathogenesis, classification, and management. Cureus 2022;14(10):e30330. [CrossRef]
[Pubmed]

2.
Carrión-Barberà I, Salman-Monte TC, Vílchez-Oya F, Monfort J. Neuropsychiatric involvement in systemic lupus erythematosus: A review. Autoimmun Rev 2021;20(4):102780. [CrossRef]
[Pubmed]

3.
Dhillon S. Telitacicept: First approval. Drugs 2021;81(14):1671–5. [CrossRef]
[Pubmed]

4.
Wang X, Zhang Q, Luo S, Zhang H, Lu Q, Long H. Advances in therapeutic targets-related study on systemic lupus erythematosus. Zhong Nan Da Xue Xue Bao Yi Xue Ban 2021;46(11):1267–75. [CrossRef]
[Pubmed]

5.
Schwartz N, Stock AD, Putterman C. Neuropsychiatric lupus: New mechanistic insights and future treatment directions. Nat Rev Rheumatol 2019;15(3):137–52. [CrossRef]
[Pubmed]

6.
Ma K, Du W, Wang X, Yuan S, Cai X, Liu D, et al. Multiple functions of B cells in the pathogenesis of systemic lupus erythematosus. Int J Mol Sci 2019;20(23):6021. [CrossRef]
[Pubmed]

7.
Kil LP, Hendriks RW. Aberrant B cell selection and activation in systemic lupus erythematosus. Int Rev Immunol 2013;32(4):445–70. [CrossRef]
[Pubmed]

8.
Wen J, Stock AD, Chalmers SA, Putterman C. The role of B cells and autoantibodies in neuropsychiatric lupus. Autoimmun Rev 2016;15(9):890–5. [CrossRef]
[Pubmed]

9.
Sakai J, Akkoyunlu M. The role of BAFF system molecules in host response to pathogens. Clin Microbiol Rev 2017;30(4):991–1014. [CrossRef]
[Pubmed]

10.
Möckel T, Basta F, Weinmann-Menke J, Schwarting A. B cell activating factor (BAFF): Structure, functions, autoimmunity and clinical implications in Systemic Lupus Erythematosus (SLE). Autoimmun Rev 2021;20(2):102736. [CrossRef]
[Pubmed]

11.
Hopia L, Thangarajh M, Khademi M, Laveskog A, Wallström E, Svenungsson E, et al. Cerebrospinal fluid levels of a proliferation-inducing ligand (APRIL) are increased in patients with neuropsychiatric systemic lupus erythematosus. Scand J Rheumatol 2011;40(5):363–72. [CrossRef]
[Pubmed]

12.
Salazar-Camarena DC, Ortiz-Lazareno PC, Cruz A, Oregon-Romero E, Machado-Contreras JR, Muñoz-Valle JF, et al. Association of BAFF, APRIL serum levels, BAFF-R, TACI and BCMA expression on peripheral B-cell subsets with clinical manifestations in systemic lupus erythematosus. Lupus 2016;25(6):582–92. [CrossRef]
[Pubmed]

13.
Furie RA, Wallace DJ, Aranow C, Fettiplace J, Wilson B, Mistry P, et al. Long-term safety and efficacy of belimumab in patients with systemic lupus erythematosus: A continuation of a seventy-six-week phase III parent study in the United States. Arthritis Rheumatol 2018;70(6):868–77. [CrossRef]
[Pubmed]

14.
Xie W, Huang H, Zhan S, Zhang Z. Risk of psychiatric disorders and all-cause mortality with belimumab therapy in patients with systemic lupus erythematosus: A meta-analysis of randomised controlled trials. Lupus Sci Med 2021;8(1):e000534. [CrossRef]
[Pubmed]

15.
Chen F, Zheng Y, Chen X, Wen Z, Xu Y, Yang J, et al. Belimumab in childhood systemic lupus erythematosus: A review of available data. Front Immunol 2022;13:940416. [CrossRef]
[Pubmed]

16.
Deijns SJ, Broen JCA, Kruyt ND, Schubart CD, Andreoli L, Tincani A, et al. The immunologic etiology of psychiatric manifestations in systemic lupus erythematosus: A narrative review on the role of the blood brain barrier, antibodies, cytokines and chemokines. Autoimmun Rev 2020;19(8):102592. [CrossRef]
[Pubmed]

17.
George-Chandy A, Trysberg E, Eriksson K. Raised intrathecal levels of APRIL and BAFF in patients with systemic lupus erythematosus: Relationship to neuropsychiatric symptoms. Arthritis Res Ther 2008;10(4):R97. [CrossRef]
[Pubmed]

18.
Cornelis R, Chang HD, Radbruch A. Keeping up with the stress of antibody production: BAFF and APRIL maintain memory plasma cells. Curr Opin Immunol 2021;71:97–102. [CrossRef]
[Pubmed]

19.
Jin HZ, Li YJ, Wang X, Li Z, Ma B, Niu L, et al. Efficacy and safety of telitacicept in patients with systemic lupus erythematosus: A multicentre, retrospective, real-world study. Lupus Sci Med 2023;10(2):e001074. [CrossRef]
[Pubmed]

20.
Xie W, Chen P, Luo L. Case report: A patient presenting primarily with psychosis of NPSLE treated with telitacicept, corticosteroids, and immunosuppressants. Front Immunol 2025;16:1626721. [CrossRef]
[Pubmed]

SUPPORTING INFORMATION
Author Contributions
Wei Zhang - Conception of the work, Design of the work, Acquisition of data, Analysis of data, Drafting the work, Revising the work critically for important intellectual content, Final approval of the version to be published, Agree to be accountable for all aspects of the work in ensuring that questions related to the accuracy or integrity of any part of the work are appropriately investigated and resolved.
Yawen Zheng - Conception of the work, Design of the work, Acquisition of data, Analysis of data, Drafting the work, Revising the work critically for important intellectual content, Final approval of the version to be published, Agree to be accountable for all aspects of the work in ensuring that questions related to the accuracy or integrity of any part of the work are appropriately investigated and resolved.
Xixi Chen - Conception of the work, Design of the work, Acquisition of data, Analysis of data, Drafting the work, Revising the work critically for important intellectual content, Final approval of the version to be published, Agree to be accountable for all aspects of the work in ensuring that questions related to the accuracy or integrity of any part of the work are appropriately investigated and resolved.
Li Long - Conception of the work, Design of the work, Acquisition of data, Analysis of data, Drafting the work, Revising the work critically for important intellectual content, Final approval of the version to be published, Agree to be accountable for all aspects of the work in ensuring that questions related to the accuracy or integrity of any part of the work are appropriately investigated and resolved.
Guaranter of SubmissionThe corresponding author is the guarantor of submission.
Source of SupportNone
Consent StatementWritten informed consent was obtained from the patient for publication of this article.
Data AvailabilityAll relevant data are within the paper and its Supporting Information files.
Conflict of InterestAuthors declare no conflict of interest.
Copyright© 2026 Wei Zhang et al. This article is distributed under the terms of Creative Commons Attribution License which permits unrestricted use, distribution and reproduction in any medium provided the original author(s) and original publisher are properly credited. Please see the copyright policy on the journal website for more information.